 CHEMBL2337271 BDBM50429790 Donepezil Hydrochloride Monohydrate
CHEMBL2337271 BDBM50429790 Donepezil Hydrochloride Monohydrate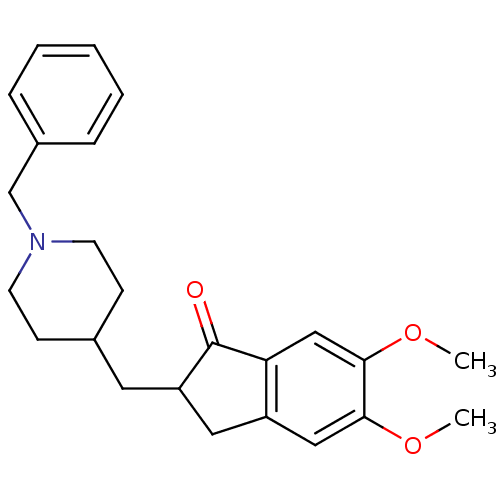 US8999994, Donepezil E2020 2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-2,3-dihydro-1H-inden-1-one BDBM8960 (+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-indan-1-one Aricept Aricept odt CHEMBL2337271 US9586925, Donepezil CHEMBL1678 Donepzil CHEMBL502 Donepezil US9346818, DPH US9663465, Donepezil DONEPEZIL HYDROCHLORIDE
US8999994, Donepezil E2020 2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-2,3-dihydro-1H-inden-1-one BDBM8960 (+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-indan-1-one Aricept Aricept odt CHEMBL2337271 US9586925, Donepezil CHEMBL1678 Donepzil CHEMBL502 Donepezil US9346818, DPH US9663465, Donepezil DONEPEZIL HYDROCHLORIDE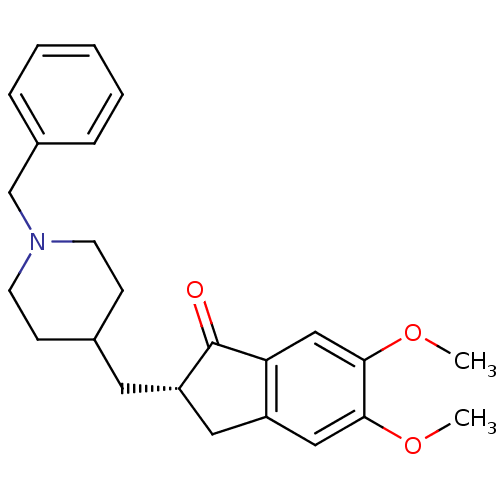 DONEPEZIL (2S)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one CHEMBL108726 (S)-donepezil BDBM50037176
DONEPEZIL (2S)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one CHEMBL108726 (S)-donepezil BDBM50037176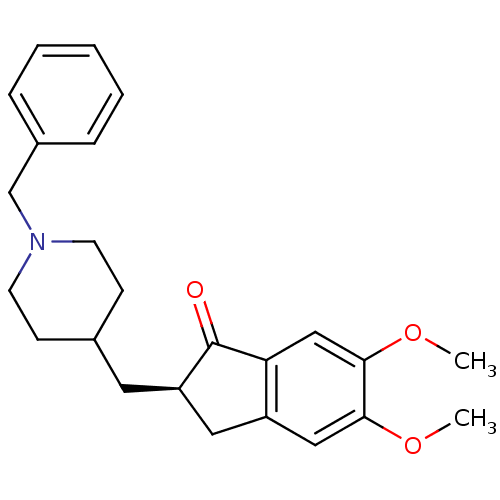 (R)-donepezil (2R)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one BDBM50037187
(R)-donepezil (2R)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one BDBM50037187 CHEMBL1678 BDBM50029923 Donepezil E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG DONEPEZIL HYDROCHLORIDE 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt
CHEMBL1678 BDBM50029923 Donepezil E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG DONEPEZIL HYDROCHLORIDE 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt CHEMBL1678 Donepezil BDBM50342148 E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG Donepezil hydrochloride 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt
CHEMBL1678 Donepezil BDBM50342148 E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG Donepezil hydrochloride 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt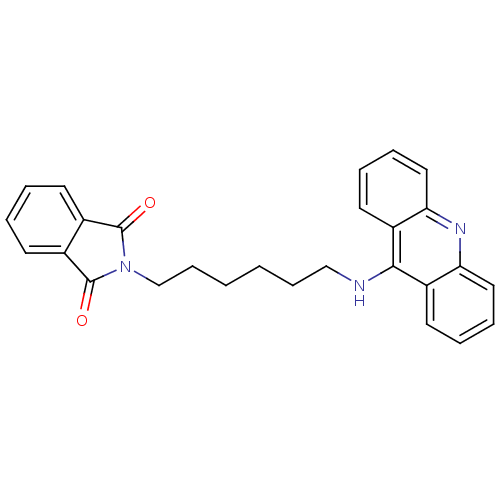 2-[6-(acridin-9-ylamino)hexyl]-2,3-dihydro-1H-isoindole-1,3-dione BDBM9070 2-[6-(Acridin-9-ylamino)-hexyl]-isoindole-1,3-dione Donepezil-tacrine hybrid 19
2-[6-(acridin-9-ylamino)hexyl]-2,3-dihydro-1H-isoindole-1,3-dione BDBM9070 2-[6-(Acridin-9-ylamino)-hexyl]-isoindole-1,3-dione Donepezil-tacrine hybrid 19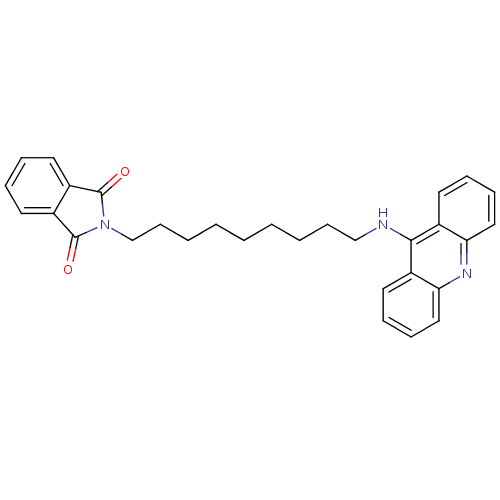 2-[9-(acridin-9-ylamino)nonyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 22 2-[9-(Acridin-9-ylamino)-nonyl]-isoindole-1,3-dione BDBM9073
2-[9-(acridin-9-ylamino)nonyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 22 2-[9-(Acridin-9-ylamino)-nonyl]-isoindole-1,3-dione BDBM9073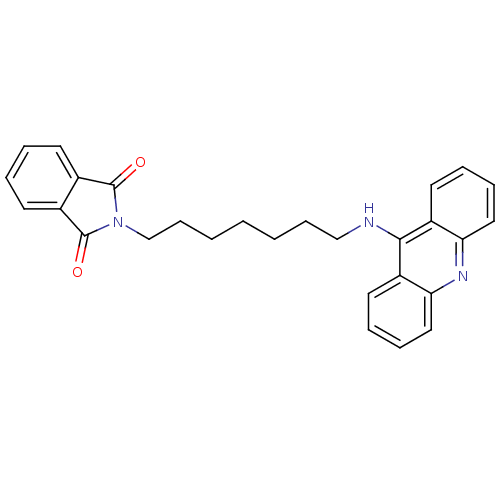 BDBM9071 2-[7-(acridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 20 2-[7-(Acridin-9-ylamino)-heptyl]-isoindole-1,3-dione
BDBM9071 2-[7-(acridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 20 2-[7-(Acridin-9-ylamino)-heptyl]-isoindole-1,3-dione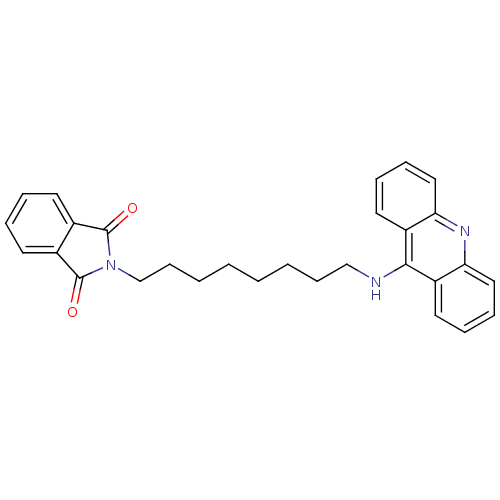 BDBM9072 2-[8-(Acridin-9-ylamino)-octyl]-isoindole-1,3-dione 2-[8-(acridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 21
BDBM9072 2-[8-(Acridin-9-ylamino)-octyl]-isoindole-1,3-dione 2-[8-(acridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 21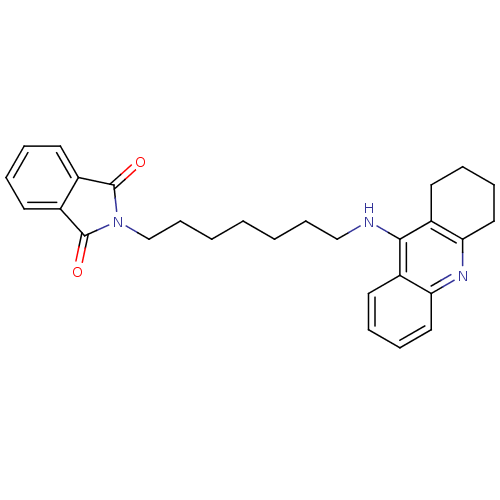 2-[7-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-heptyl]-isoindole-1,3-dione 2-[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 17 BDBM9068
2-[7-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-heptyl]-isoindole-1,3-dione 2-[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 17 BDBM9068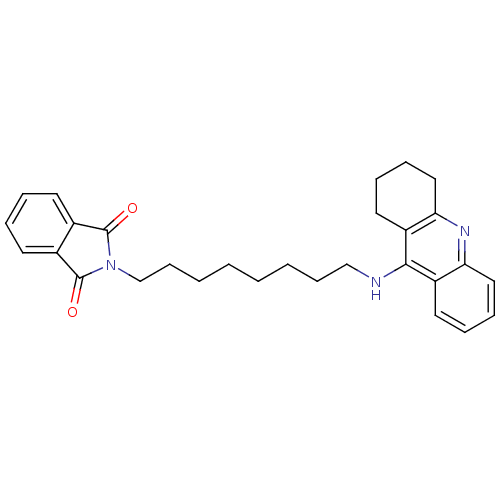 2-[8-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-octyl]-isoindole-1,3-dione BDBM9069 2-[8-(1,2,3,4-tetrahydroacridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 18
2-[8-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-octyl]-isoindole-1,3-dione BDBM9069 2-[8-(1,2,3,4-tetrahydroacridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 18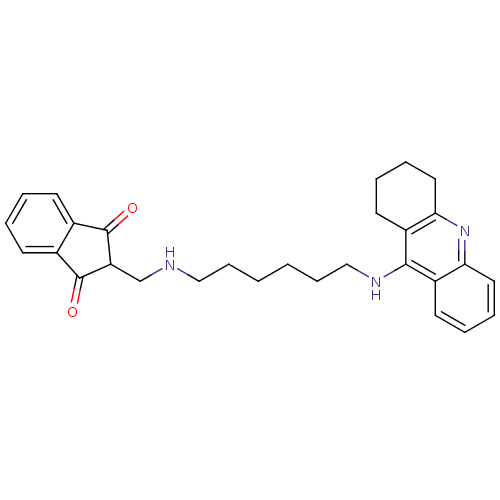 2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-1H-indene-1,3(2H)-dione Donepezil-tacrine hybrid 9 2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-indene-1,3-dione BDBM9064
2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-1H-indene-1,3(2H)-dione Donepezil-tacrine hybrid 9 2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-indene-1,3-dione BDBM9064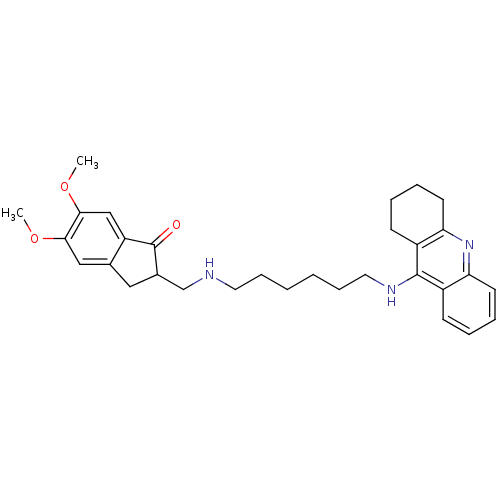 5,6-Dimethoxy-2-{[6-(1,2,3,4-tetrahydro-acridin-9-ylamino)-hexylamino]-methyl}-indan-1-one 5,6-dimethoxy-2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-inden-1-one Donepezil-tacrine hybrid 7 BDBM9062
5,6-Dimethoxy-2-{[6-(1,2,3,4-tetrahydro-acridin-9-ylamino)-hexylamino]-methyl}-indan-1-one 5,6-dimethoxy-2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-inden-1-one Donepezil-tacrine hybrid 7 BDBM9062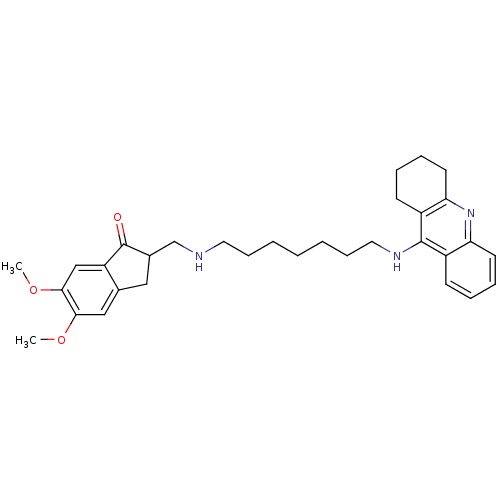 CHEMBL484306 5,6-dimethoxy-2-({[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]amino}methyl)-2,3-dihydro-1H-inden-1-one 5,6-Dimethoxy-2-{[7-(1,2,3,4-tetrahydro-acridin-9-ylamino)-heptylamino]-methyl}-indan-1-one Donepezil-tacrine hybrid 8 BDBM9063
CHEMBL484306 5,6-dimethoxy-2-({[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]amino}methyl)-2,3-dihydro-1H-inden-1-one 5,6-Dimethoxy-2-{[7-(1,2,3,4-tetrahydro-acridin-9-ylamino)-heptylamino]-methyl}-indan-1-one Donepezil-tacrine hybrid 8 BDBM9063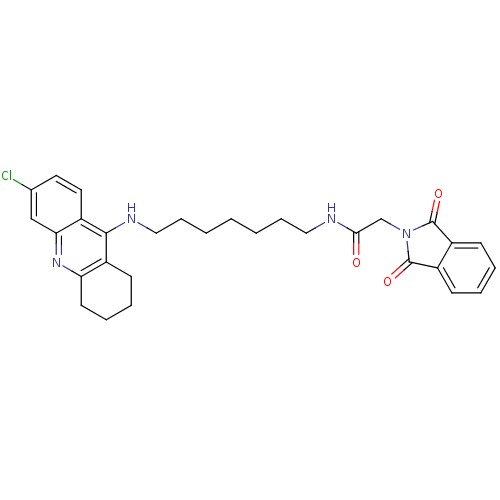 N-[7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-heptyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide N-{7-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]heptyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 11 BDBM9066
N-[7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-heptyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide N-{7-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]heptyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 11 BDBM9066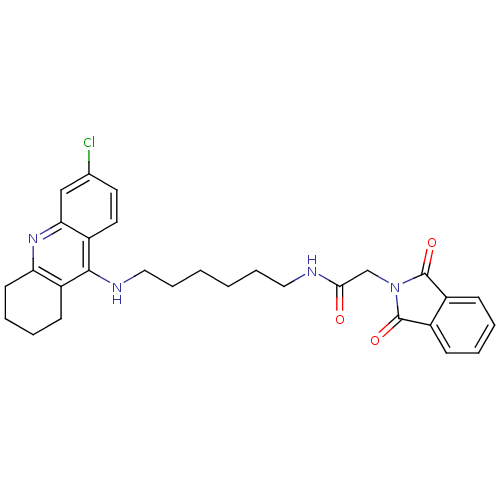 N-{6-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]hexyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 10 N-[6-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-hexyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide BDBM9065
N-{6-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]hexyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 10 N-[6-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-hexyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide BDBM9065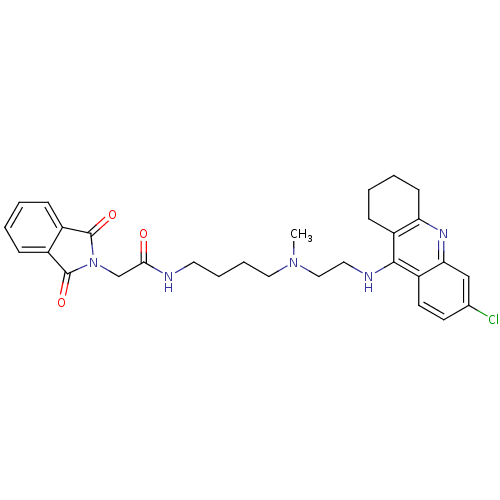 N-{4-[{2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino]butyl}-2-(1,3-dioxo-1,3-dihydro-2H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 12 BDBM9067 N-[4-({2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino)butyl]-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide
N-{4-[{2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino]butyl}-2-(1,3-dioxo-1,3-dihydro-2H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 12 BDBM9067 N-[4-({2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino)butyl]-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide
- Yerdelen, KO; Koca, M; Anil, B; Sevindik, H; Kasap, Z; Halici, Z; Turkaydin, K; Gunesacar, G Synthesis of donepezil-based multifunctional agents for the treatment of Alzheimer's disease. Bioorg Med Chem Lett 25: 5576-82 (2015)
- Saglik, BN; Ilgin, S; �zkay, Y Synthesis of new donepezil analogues and investigation of their effects on cholinesterase enzymes. Eur J Med Chem 124: 1026-1040 (2016)
- Li, Q; He, S; Chen, Y; Feng, F; Qu, W; Sun, H Donepezil-based multi-functional cholinesterase inhibitors for treatment of Alzheimer's disease. Eur J Med Chem 158: 463-477 (2018)
- Wang, ZM; Cai, P; Liu, QH; Xu, DQ; Yang, XL; Wu, JJ; Kong, LY; Wang, XB Rational modification of donepezil as multifunctional acetylcholinesterase inhibitors for the treatment of Alzheimer's disease. Eur J Med Chem 123: 282-297 (2016)
- Omran, Z; Stiebing, S; Godard, AM; Sopkova-De Oliveira-Santos, J; Dallemagne, P Synthesis and biological evaluation of new donepezil-like Thiaindanones as AChE inhibitors. J Enzyme Inhib Med Chem 23: 696-703 (2008)
- Combination of NSAIDs with donepezil as multi-target directed ligands for the treatment of Alzheimer's disease.
- Costanzo, P; Cariati, L; Desiderio, D; Sgammato, R; Lamberti, A; Arcone, R; Salerno, R; Nardi, M; Masullo, M; Oliverio, M Design, Synthesis, and Evaluation of Donepezil-Like Compounds as AChE and BACE-1 Inhibitors. ACS Med Chem Lett 7: 470-5 (2016)
- Hu, Y; Zhang, J; Chandrashankra, O; Ip, FC; Ip, NY Design, synthesis and evaluation of novel heterodimers of donepezil and huperzine fragments as acetylcholinesterase inhibitors. Bioorg Med Chem 21: 676-83 (2013)
- Wu, MY; Esteban, G; Brogi, S; Shionoya, M; Wang, L; Campiani, G; Unzeta, M; Inokuchi, T; Butini, S; Marco-Contelles, J Donepezil-like multifunctional agents: Design, synthesis, molecular modeling and biological evaluation. Eur J Med Chem 121: 864-879 (2016)
- McEneny-King, A; Edginton, AN; Rao, PP Investigating the binding interactions of the anti-Alzheimer's drug donepezil with CYP3A4 and P-glycoprotein. Bioorg Med Chem Lett 25: 297-301 (2014)
- Zhu, G; Wang, K; Shi, J; Zhang, P; Yang, D; Fan, X; Zhang, Z; Liu, W; Sang, Z The development of 2-acetylphenol-donepezil hybrids as multifunctional agents for the treatment of Alzheimer's disease. Bioorg Med Chem Lett 29: (2019)
- Alonso, D; Dorronsoro, I; Rubio, L; Munoz, P; Garcia-Palomero, E; Del Monte, M; Bidon-Chanal, A; Orozco, M; Luque, FJ; Castro, A; Medina, M; Martinez, A Donepezil-tacrine hybrid related derivatives as new dual binding site inhibitors of AChE. Bioorg Med Chem 13: 6588-97 (2005)
- Camps, P; Formosa, X; Galdeano, C; Gómez, T; Muñoz-Torrero, D; Scarpellini, M; Viayna, E; Badia, A; Clos, MV; Camins, A; Pallàs, M; Bartolini, M; Mancini, F; Andrisano, V; Estelrich, J; Lizondo, M; Bidon-Chanal, A; Luque, FJ Novel donepezil-based inhibitors of acetyl- and butyrylcholinesterase and acetylcholinesterase-induced beta-amyloid aggregation. J Med Chem 51: 3588-98 (2008)
- van Greunen, DG; Cordier, W; Nell, M; van der Westhuyzen, C; Steenkamp, V; Panayides, JL; Riley, DL Targeting Alzheimer's disease by investigating previously unexplored chemical space surrounding the cholinesterase inhibitor donepezil. Eur J Med Chem 127: 671-690 (2017)
- Wang, XB; Yin, FC; Huang, M; Jiang, N; Lan, JS; Kong, LY Chromone and donepezil hybrids as new multipotent cholinesterase and monoamine oxidase inhibitors for the potential treatment of Alzheimer's disease. RSC Med Chem 11: 225-233 (2020)
- Dias, KS; de Paula, CT; Dos Santos, T; Souza, IN; Boni, MS; Guimar�es, MJ; da Silva, FM; Castro, NG; Neves, GA; Veloso, CC; Coelho, MM; de Melo, IS; Giusti, FC; Giusti-Paiva, A; da Silva, ML; Dardenne, LE; Guedes, IA; Pruccoli, L; Morroni, F; Tarozzi, A; Viegas, C Design, synthesis and evaluation of novel feruloyl-donepezil hybrids as potential multitarget drugs for the treatment of Alzheimer's disease. Eur J Med Chem 130: 440-457 (2017)
- Cristancho Ortiz, CJ; de Freitas Silva, M; Pruccoli, L; Fonseca Nadur, N; de Azevedo, LL; K�mmerle, AE; Guedes, IA; Dardenne, LE; Leomil Coelho, LF; Guimar�es, MJ; da Silva, FMR; Castro, N; Gontijo, VS; Rojas, VCT; de Oliveira, MK; Vilela, FC; Giusti-Paiva, A; Barbosa, G; Lima, LM; Pinheiro, GB; Veras, LG; Mortari, MR; Tarozzi, A; Viegas, C Design, synthesis, and biological evaluation of new thalidomide-donepezil hybrids as neuroprotective agents targeting cholinesterases and neuroinflammation. RSC Med Chem 13: 568-584 (2022)
- Yan, J; Hu, J; Liu, A; He, L; Li, X; Wei, H Design, synthesis, and evaluation of multitarget-directed ligands against Alzheimer's disease based on the fusion of donepezil and curcumin. Bioorg Med Chem 25: 2946-2955 (2017)
- Cai, P; Fang, SQ; Yang, HL; Yang, XL; Liu, QH; Kong, LY; Wang, XB Donepezil-butylated hydroxytoluene (BHT) hybrids as Anti-Alzheimer's disease agents with cholinergic, antioxidant, and neuroprotective properties. Eur J Med Chem 157: 161-176 (2018)
- Estrada Valencia, M; Herrera-Arozamena, C; de Andr�s, L; P�rez, C; Morales-Garc�a, JA; P�rez-Castillo, A; Ramos, E; Romero, A; Vi�a, D; Y��ez, M; Laurini, E; Pricl, S; Rodr�guez-Franco, MI Neurogenic and neuroprotective donepezil-flavonoid hybrids with sigma-1 affinity and inhibition of key enzymes in Alzheimer's disease. Eur J Med Chem 156: 534-553 (2018)
- Brunetti, L; Leuci, R; Carrieri, A; Catto, M; Occhineri, S; Vinci, G; Gambacorta, L; Baltrukevich, H; Chaves, S; Laghezza, A; Altomare, CD; Tortorella, P; Santos, MA; Loiodice, F; Piemontese, L Structure-based design of novel donepezil-like hybrids for a multi-target approach to the therapy of Alzheimer's disease. Eur J Med Chem 237: (2022)
- Luo, Z; Sheng, J; Sun, Y; Lu, C; Yan, J; Liu, A; Luo, HB; Huang, L; Li, X Synthesis and evaluation of multi-target-directed ligands against Alzheimer's disease based on the fusion of donepezil and ebselen. J Med Chem 56: 9089-99 (2013)
- Wang, J; Wang, ZM; Li, XM; Li, F; Wu, JJ; Kong, LY; Wang, XB Synthesis and evaluation of multi-target-directed ligands for the treatment of Alzheimer's disease based on the fusion of donepezil and melatonin. Bioorg Med Chem 24: 4324-4338 (2016)
- Xie, SS; Lan, JS; Wang, X; Wang, ZM; Jiang, N; Li, F; Wu, JJ; Wang, J; Kong, LY Design, synthesis and biological evaluation of novel donepezil-coumarin hybrids as multi-target agents for the treatment of Alzheimer's disease. Bioorg Med Chem 24: 1528-39 (2016)
- Gabr, MT; Abdel-Raziq, MS Structure-based design, synthesis, and evaluation of structurally rigid donepezil analogues as dual AChE and BACE-1 inhibitors. Bioorg Med Chem Lett 28: 2910-2913 (2018)
- Andreani, A; Cavalli, A; Granaiola, M; Guardigli, M; Leoni, A; Locatelli, A; Morigi, R; Rambaldi, M; Recanatini, M; Roda, A Synthesis and screening for antiacetylcholinesterase activity of (1-benzyl-4-oxopiperidin-3-ylidene)methylindoles and -pyrroles related to donepezil. J Med Chem 44: 4011-4 (2001)
- Mishra, CB; Kumari, S; Manral, A; Prakash, A; Saini, V; Lynn, AM; Tiwari, M Design, synthesis, in-silico and biological evaluation of novel donepezil derivatives as multi-target-directed ligands for the treatment of Alzheimer's disease. Eur J Med Chem 125: 736-750 (2017)
- Wang, L; Esteban, G; Ojima, M; Bautista-Aguilera, OM; Inokuchi, T; Moraleda, I; Iriepa, I; Samadi, A; Youdim, MB; Romero, A; Soriano, E; Herrero, R; Fern�ndez Fern�ndez, AP; Ricardo-Mart�nez-Murillo, na; Marco-Contelles, J; Unzeta, M Donepezil + propargylamine + 8-hydroxyquinoline hybrids as new multifunctional metal-chelators, ChE and MAO inhibitors for the potential treatment of Alzheimer's disease. Eur J Med Chem 80: 543-61 (2014)
- Li, X; Li, J; Huang, Y; Gong, Q; Fu, Y; Xu, Y; Huang, J; You, H; Zhang, D; Zhang, D; Mao, F; Zhu, J; Wang, H; Zhang, H; Li, J The novel therapeutic strategy of vilazodone-donepezil chimeras as potent triple-target ligands for the potential treatment of Alzheimer's disease with comorbid depression. Eur J Med Chem 229: (2022)
- Peauger, L; Azzouz, R; Gembus, V; T�ntas, ML; Sopkov�-de Oliveira Santos, J; Bohn, P; Papamica�l, C; Levacher, V Donepezil-Based Central Acetylcholinesterase Inhibitors by Means of a"Bio-Oxidizable" Prodrug Strategy: Design, Synthesis, and in Vitro Biological Evaluation. J Med Chem 60: 5909-5926 (2017)
- Lan, JS; Zhang, T; Liu, Y; Yang, J; Xie, SS; Liu, J; Miao, ZY; Ding, Y Design, synthesis and biological activity of novel donepezil derivatives bearing N-benzyl pyridinium moiety as potent and dual binding site acetylcholinesterase inhibitors. Eur J Med Chem 133: 184-196 (2017)
- Monjas, L; Arce, MP; Le�n, R; Egea, J; P�rez, C; Villarroya, M; L�pez, MG; Gil, C; Conde, S; Rodr�guez-Franco, MI Enzymatic and solid-phase synthesis of new donepezil-based L- and d-glutamic acid derivatives and their pharmacological evaluation in models related to Alzheimer's disease and cerebral ischemia. Eur J Med Chem 130: 60-72 (2017)
- Azzouz, R; Peauger, L; Gembus, V; ?�n?a?, ML; Sopkov�-de Oliveira Santos, J; Papamica�l, C; Levacher, V Novel donepezil-like N-benzylpyridinium salt derivatives as AChE inhibitors and their corresponding dihydropyridine "bio-oxidizable" prodrugs: Synthesis, biological evaluation and structure-activity relationship. Eur J Med Chem 145: 165-190 (2018)
- Dias Viegas, FP; de Freitas Silva, M; Divino da Rocha, M; Castelli, MR; Riquiel, MM; Machado, RP; Vaz, SM; Sim�es de Lima, LM; Mancini, KC; Marques de Oliveira, PC; Morais, �P; Gontijo, VS; da Silva, FMR; D'Alincourt da Fonseca Pe�anha, D; Castro, NG; Neves, GA; Giusti-Paiva, A; Vilela, FC; Orlandi, L; Camps, I; Veloso, MP; Leomil Coelho, LF; Ionta, M; Ferreira-Silva, G�; Pereira, RM; Dardenne, LE; Guedes, IA; de Oliveira Carneiro Junior, W; Quaglio Bellozi, PM; Pinheiro de Oliveira, AC; Ferreira, FF; Pruccoli, L; Tarozzi, A; Viegas, C Design, synthesis and pharmacological evaluation of N-benzyl-piperidinyl-aryl-acylhydrazone derivatives as donepezil hybrids: Discovery of novel multi-target anti-alzheimer prototype drug candidates. Eur J Med Chem 147: 48-65 (2018)
- Bautista-Aguilera, OM; Esteban, G; Bolea, I; Nikolic, K; Agbaba, D; Moraleda, I; Iriepa, I; Samadi, A; Soriano, E; Unzeta, M; Marco-Contelles, J Design, synthesis, pharmacological evaluation, QSAR analysis, molecular modeling and ADMET of novel donepezil-indolyl hybrids as multipotent cholinesterase/monoamine oxidase inhibitors for the potential treatment of Alzheimer's disease. Eur J Med Chem 75: 82-95 (2014)
- Bolea, I; Juárez-Jiménez, J; de Los Ríos, C; Chioua, M; Pouplana, R; Luque, FJ; Unzeta, M; Marco-Contelles, J; Samadi, A Synthesis, biological evaluation, and molecular modeling of donepezil and N-[(5-(benzyloxy)-1-methyl-1H-indol-2-yl)methyl]-N-methylprop-2-yn-1-amine hybrids as new multipotent cholinesterase/monoamine oxidase inhibitors for the treatment of Alzheimer's disease. J Med Chem 54: 8251-70 (2011)
- AChE and BChE Inhibitory Activities Newly synthesized coumarin thioureas were tested against electric eel AChE and horse serum BChE. The cholinesterase inhibitory activity was measured using standard protocol. Donepezil and neostigmine were used as standard references in the assay. The compounds were initially tested against theseenzymes at 1 mM concentration.
- Cholinesterase Activity Assay Reactions were performed in a mediumcontaining substrate (0.05-0.4 mM) combined with 0.125 mM DTNB in 100 mM 3-(N-morpholino)propanesulfonic acid buffer, pH 8.0 at 25 °C, initiated by the addition of enzyme, and monitored spectrophotometrically at 412 nm in a UV-visible 1700 Shimadzu PC spectrophotometer. The activity was determined by measuring the increase in absorbance at 412 nm for 7 min (εTNB = 14.2 mM^-1 cm^-1). Activities of the treated samples were compared to control to estimate percentage of inhibition (I%). Dose-response curves were plotted using the GraphPad Prism 5 software and IC50 values were determined. Data are expressed as mean ± SEM. An enzyme kinetic assay was performed for compound 2 at different butyrylthiocholine (BTC) concentrations (0.05-0.3 mM). Donepezil-HCl (Sigma) was tested as a reference compound.
- Inhibition Assay Acetylcholinesterase extracted from human erythrocytes (buffered aqueous solution, ≧500 units/mg, Sigma Aldrich) is diluted in a 20 mM HEPES buffer pH8, 0.1% Triton X-100, to obtain a standard solution with 2.5 units of enzyme activity/ml. 100 μL of a 0.3 mM 5,5-dithiobis(2-nitrobenzoic acid) (DTNB) solution in a pH 7.4 phosphate buffer are introduced into a 96-well plate, followed by 50 μL of compound to be tested in solution in DMSO and 50 μL of enzyme solution. After 5 min of preincubation, the reaction is initiated by injecting 50 μL of 10 mM acetylthiocholine iodide solution. Hydrolysis of the acetylthiocholine is followed by the formation of the anion 5-thio-2-nitrobenzoate, produced by the reaction of the DTNB with the thiocholine released by the enzymatic hydrolysis of the acetylthiocholine, by means of a microplate reader (TECAN Infinite M200, Lyon, France) at a wavelength of 412 nm per minute for 10 minutes. Donepezil is used as a reference product.
- Anticholinesterase Enzyme Inhibition Assay The in vitro AChE inhibitory activity was measured using the methods described earlier [Biochem. Pharmacol. 7:88-95]. Briefly, stock solutions (1mg/mL) of test compounds were prepared using DMSO. Working solutions (0.01-100�g/mL) were prepared by serial dilutions. The various concentrations of test compounds (10 �L) were pre-incubated with sodium phosphate buffer (0.1M; pH 8.0; 150 �L), and AChE solution (0.1 U/mL; 20 �L) for 15 min at 25 C. The reaction was initiated by addition of DTNB (10 mM; 10 �L) and ATChI (14 mM; 10 �L). The reaction mixture was mixed using a cyclomixer and incubated for 10min at room temperature. The absorbance was measured using a microplate reader at 410 nm wavelength against the blank reading containing 10�L DMSO instead of test compound. The % inhibition was calculated using the formula described in Eq. (1) and the IC50 was calculated. Donepezil (0.01-100�g/mL) was used as the positive control.
- Cholinesterase Inhibition Assay Suitable agents like donepezil hydrochloride and tacrine hydrochloride hydrate were used as standards for this study. Buffer solution (50 mM Tris-HCl, pH 8.0, 0.1 M NaCl, 0.02 M MgCl2 6H2O) was used to dilute the stock solutions of the test compounds dissolved in minimum volume of DMSO (1%). First of all 50 �L of AChE (0.22 U/mL prepared in 50 mM Tris-HCl, pH 8.0, 0.1% w/v bovine serum albumin, BSA) or 50 �L of BuChE (0.06 U/mL prepared in 50 mM Tris-HCl, pH 8.0, 0.1% w/v BSA) and 10 �L of different concentrations of the test and standard compounds (0.001-100 �M) were incubated in 96-well plates at room temperature for 30 min. Further, 30 �L of the substrate viz. Acetylthiocholine iodide [ATCI (15 mM)] or butyrlthiocholine iodide [BTCI (15 mM)] was added and incubated for 30 min. Finally 160 �L DTNB (1.5 mM) was added and absorbance was measured at 415 nm wavelength.
- AChE/BChE Inhibitory Assay AChE and BChE inhibitory assay was carried out by following Ellman's methodology [Ellman et al., Biochem. Pharmacol., 7:88-95] using AChE (Electric eel type-VI-S, Sigma-Aldrich GmbH USA, code 1001596210), BChE (Equine serum Lyophilized Sigma-Aldrich GmbH USA, code 101292670) and DTNB (Sigma-Aldrich Germany, code 101261619), which produced colored product (5-thio-2-nitrobenzoate) whose concentration can be measured by the increase in absorbance at 412 nm using mQuant microplate spectrophotometer (MQX200, BioTek USA). Other reagents, like Acetylthiocholine iodide (Sigma-Aldrich UK, code 101303874), Butyrylthiocholine Iodide (Sigma-Aldrich Switzerland, code 101334643) were employed. Galantamine hydrobromide Lycoris Sp. (Sigma-Aldrich France, code G1660) and Donepezil were used as reference drugs. Stock solution of the synthesized quinazoline was prepared with 0.1 M phosphate buffer (KH2PO4/K2HPO4) having of pH 8.0. Appropriate amount of DTNB (Ellman's reagent), quinazoline compounds, 0.03 U/ml of enzymes (AChE and BChE) were reacted by pre-incubating at 30 °C for 10 min and then further incubating for 15 min after addition of 1 mM ATCI or BTCI.
 CHEMBL2337271 BDBM50429790 Donepezil Hydrochloride Monohydrate
CHEMBL2337271 BDBM50429790 Donepezil Hydrochloride Monohydrate US8999994, Donepezil E2020 2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-2,3-dihydro-1H-inden-1-one BDBM8960 (+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-indan-1-one Aricept Aricept odt CHEMBL2337271 US9586925, Donepezil CHEMBL1678 Donepzil CHEMBL502 Donepezil US9346818, DPH US9663465, Donepezil DONEPEZIL HYDROCHLORIDE
US8999994, Donepezil E2020 2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-2,3-dihydro-1H-inden-1-one BDBM8960 (+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxy-indan-1-one Aricept Aricept odt CHEMBL2337271 US9586925, Donepezil CHEMBL1678 Donepzil CHEMBL502 Donepezil US9346818, DPH US9663465, Donepezil DONEPEZIL HYDROCHLORIDE DONEPEZIL (2S)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one CHEMBL108726 (S)-donepezil BDBM50037176
DONEPEZIL (2S)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one CHEMBL108726 (S)-donepezil BDBM50037176 (R)-donepezil (2R)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one BDBM50037187
(R)-donepezil (2R)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimethoxyindan-1-one BDBM50037187 CHEMBL1678 BDBM50029923 Donepezil E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG DONEPEZIL HYDROCHLORIDE 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt
CHEMBL1678 BDBM50029923 Donepezil E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG DONEPEZIL HYDROCHLORIDE 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt CHEMBL1678 Donepezil BDBM50342148 E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG Donepezil hydrochloride 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt
CHEMBL1678 Donepezil BDBM50342148 E-2020 2-(1-Benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indan-1-one; hydrochloride Aricept BNAG Donepezil hydrochloride 2-((1-benzylpiperidin-4-yl)methyl)-5,6-dimethoxy-2,3-dihydroinden-1-one hydrochloride Aricept odt 2-[6-(acridin-9-ylamino)hexyl]-2,3-dihydro-1H-isoindole-1,3-dione BDBM9070 2-[6-(Acridin-9-ylamino)-hexyl]-isoindole-1,3-dione Donepezil-tacrine hybrid 19
2-[6-(acridin-9-ylamino)hexyl]-2,3-dihydro-1H-isoindole-1,3-dione BDBM9070 2-[6-(Acridin-9-ylamino)-hexyl]-isoindole-1,3-dione Donepezil-tacrine hybrid 19 2-[9-(acridin-9-ylamino)nonyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 22 2-[9-(Acridin-9-ylamino)-nonyl]-isoindole-1,3-dione BDBM9073
2-[9-(acridin-9-ylamino)nonyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 22 2-[9-(Acridin-9-ylamino)-nonyl]-isoindole-1,3-dione BDBM9073 BDBM9071 2-[7-(acridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 20 2-[7-(Acridin-9-ylamino)-heptyl]-isoindole-1,3-dione
BDBM9071 2-[7-(acridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 20 2-[7-(Acridin-9-ylamino)-heptyl]-isoindole-1,3-dione BDBM9072 2-[8-(Acridin-9-ylamino)-octyl]-isoindole-1,3-dione 2-[8-(acridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 21
BDBM9072 2-[8-(Acridin-9-ylamino)-octyl]-isoindole-1,3-dione 2-[8-(acridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 21 2-[7-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-heptyl]-isoindole-1,3-dione 2-[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 17 BDBM9068
2-[7-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-heptyl]-isoindole-1,3-dione 2-[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 17 BDBM9068 2-[8-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-octyl]-isoindole-1,3-dione BDBM9069 2-[8-(1,2,3,4-tetrahydroacridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 18
2-[8-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-octyl]-isoindole-1,3-dione BDBM9069 2-[8-(1,2,3,4-tetrahydroacridin-9-ylamino)octyl]-2,3-dihydro-1H-isoindole-1,3-dione Donepezil-tacrine hybrid 18 2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-1H-indene-1,3(2H)-dione Donepezil-tacrine hybrid 9 2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-indene-1,3-dione BDBM9064
2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-1H-indene-1,3(2H)-dione Donepezil-tacrine hybrid 9 2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-indene-1,3-dione BDBM9064 5,6-Dimethoxy-2-{[6-(1,2,3,4-tetrahydro-acridin-9-ylamino)-hexylamino]-methyl}-indan-1-one 5,6-dimethoxy-2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-inden-1-one Donepezil-tacrine hybrid 7 BDBM9062
5,6-Dimethoxy-2-{[6-(1,2,3,4-tetrahydro-acridin-9-ylamino)-hexylamino]-methyl}-indan-1-one 5,6-dimethoxy-2-({[6-(1,2,3,4-tetrahydroacridin-9-ylamino)hexyl]amino}methyl)-2,3-dihydro-1H-inden-1-one Donepezil-tacrine hybrid 7 BDBM9062 CHEMBL484306 5,6-dimethoxy-2-({[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]amino}methyl)-2,3-dihydro-1H-inden-1-one 5,6-Dimethoxy-2-{[7-(1,2,3,4-tetrahydro-acridin-9-ylamino)-heptylamino]-methyl}-indan-1-one Donepezil-tacrine hybrid 8 BDBM9063
CHEMBL484306 5,6-dimethoxy-2-({[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]amino}methyl)-2,3-dihydro-1H-inden-1-one 5,6-Dimethoxy-2-{[7-(1,2,3,4-tetrahydro-acridin-9-ylamino)-heptylamino]-methyl}-indan-1-one Donepezil-tacrine hybrid 8 BDBM9063 N-[7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-heptyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide N-{7-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]heptyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 11 BDBM9066
N-[7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-heptyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide N-{7-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]heptyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 11 BDBM9066 N-{6-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]hexyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 10 N-[6-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-hexyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide BDBM9065
N-{6-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]hexyl}-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 10 N-[6-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-hexyl]-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)acetamide BDBM9065 N-{4-[{2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino]butyl}-2-(1,3-dioxo-1,3-dihydro-2H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 12 BDBM9067 N-[4-({2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino)butyl]-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide
N-{4-[{2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino]butyl}-2-(1,3-dioxo-1,3-dihydro-2H-isoindol-2-yl)acetamide Donepezil-tacrine hybrid 12 BDBM9067 N-[4-({2-[(6-chloro-1,2,3,4-tetrahydroacridin-9-yl)amino]ethyl}(methyl)amino)butyl]-2-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)acetamide